Breast Augmentation in Bay Area
Breast Augmentation in Bay Area
Breast Augmentation Bay Area

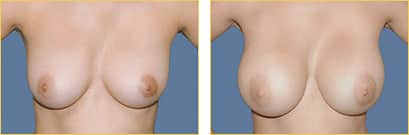
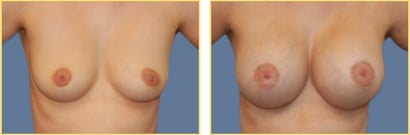
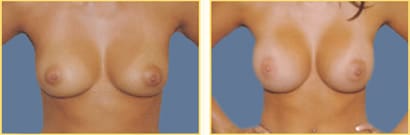
Breast augmentation surgery, also called breast implant surgery or augmentation mammoplasty, is the most popular elective cosmetic surgery performed in the United States. Breast implants can help do more than just improve breast volume and the overall size of the breasts.
Dr. Rex Moulton-Barrett offers breast augmentation surgery with implants to help women achieve lasting self-esteem and positive self-confidence in the San Francisco Bay Area, Alameda, and Brentwood. Over 280,000 women in the United States opted to undergo breast augmentation surgery this year, of which 98% of women felt the results reached or exceeded their expectations.
Breast augmentation surgeries are increasingly popular, safe, and effective. As an experienced, board-certified plastic surgeon, Dr. Moulton-Barrett is here to help you address any concerns and achieve the results you deserve.
To learn how breast augmentation surgery can benefit your body contour, request a consultation with Dr. Moulton-Barrett.
Brief Guide to Types of Breast Implants
There are three basic types of breast implants commonly used in the United States. Each breast implant can be designed to individualized patient specifications after the initial plastic surgery consultation. Patients can opt for low profile widths, high profile widths, or moderate profile widths, as well as saline implants, silicone implants, or “gummy bear” silicone implants.
1. Traditional Saline Breast Implants for for Breast Augmentation
Saline breast implants are less expensive than silicone implants. They are also recommended for people concerned about scars, as they can be placed with a smaller incision near the armpit or areola. The implant can be placed unfilled, using only a one-inch incision around the areola, for minimal visible scarring. The scar is difficult to notice, and the overall appearance of the breasts is dramatically improved. Saline-filled implants are also recommended since patients can detect any leaks before more advanced issues arise. The sterile salt water can be safely absorbed by the body in the event of breast implant rupture.
2. Structured ‘Ideal’ Saline Implants for Breast Augmentation
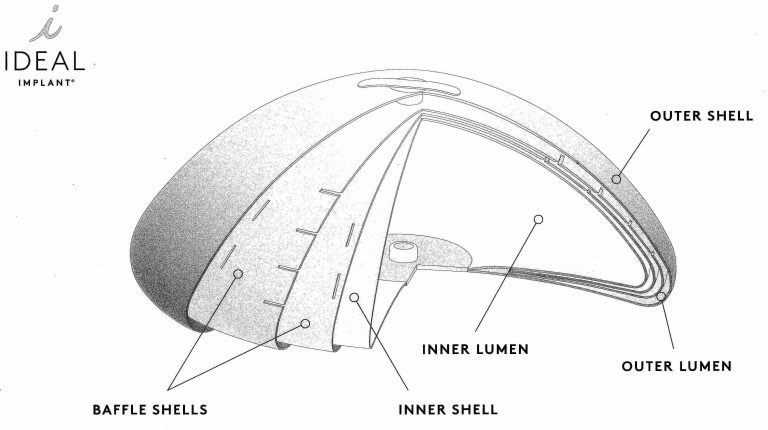

In 2015, the FDA approved for clinical use a new type of smooth saline implant called the ‘Ideal’ implant. Unlike the traditional saline implant, it is composed of an inner and larger volume chamber of sterile salt water, which is filled from the back of the implant, and a smaller and circumferential chamber which is filled from the front of the implant.
The anterior chamber has up to 3 additional leaflets of sub-chambers (called baffle shells), like the layers of an onion, which have channels to allow the saline to pass between them.
The overall effect is that the implant behaves more like natural breast tissue, which is sometimes a downside to choosing saline-filled implants. This also minimizes visible rippling and results in a consistent shape and position (including maintaining a point at the front center). Unlike a silicone implant, the ideal implant can be inserted deflated and therefore through a smaller incision.
The warranty for Ideal implants is similar to other breast implants: a lifetime replacement for both implants if there is a spontaneous rupture, and a 10-year financial aid payment of $2,400 for the cost of surgical fees as a result of replacement. Similar to other saline implants, there is no reason to replace the Ideal implant in the absence of visible deflation.
Finally, breast implant rupture is unlikely to involve both chambers, which means a deflation may be more subtle and not the devastating complete loss of breast projection that often occurs following a traditional single-chamber implant deflation.
3. Sientra “Gummy Bear” Textured Silicone Gel Implants for Breast Augmentation
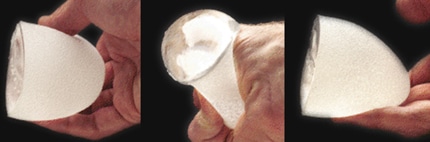
Images of a Sientra silicone implant with cohesive gel which holds form after being cut in one half
So-called gummy bear implants were recently FDA-approved for use in the United States. These new implants use a textured, viscous silicone gel for superior results. This silicone gel is thicker and more cohesive than traditional silicone gel implants, meaning it holds its shape when cut or ruptured.
This enhances their safety and minimizes the chance of implant rupture while still feeling very similar to natural breast tissue. Gummy bear breast implants provide more options for plastic surgeons and breast augmentation patients to achieve desired results with minimal scarring.
Breast Implant Placement
The placement of breast implants is critical to achieving optimal results. Placement of the breast implant under the pectoral muscle can achieve a more natural look but requires a long breast augmentation recovery. However, placing the breast implant over the pectoral muscle can protect the breast implant and reduce the risk of capsular contracture. Breast augmentation surgery scars are typically minimal and can be easily hidden. A standard one-inch incision for saline implants can be concealed by placing the incision around the areola or in the armpit. Silicone breast implants typically require larger incisions as they are pre-filled.
Silicone breast implants are often a good option for patients who have thinner chest walls, less breast tissue, or patients who are otherwise at higher risk for rippling post-surgery. Rippling is most noticeable in the lateral and lower breast poles. The right plastic surgeon can help you make an informed decision about what type of breast implant and breast surgery would be best for your individual preferences. Consult with plastic surgeon to learn more about the individualized benefits of saline breast implants, silicone breast implants, and “gummy bear” breast implants in addition to advanced methods of breast augmentation surgery that decrease the risk of scarring.
Claims that breast implants directly correlate with higher rates of breast tissue and autoimmune-related disorders are yet to be proven statistically significant. Consult with an informed and board-certified plastic surgeon about risks and individual concerns specific to medical history, lifestyle, and body type.
Lesser-Known Benefits of Breast Implants
It is widely speculated that women with breast implants are able to detect breast cancer at earlier stages per FDA guidelines. Increased awareness coupled with increased imaging can detect tumors sooner and treat cancer more effectively. It is recommended that women (with or without breast implants) receive a mammogram every one or two years after age 40. Increased accessibility to advanced imaging techniques can also help women with breast implants address saline implant leaks or silicone breast implant rupture early.
Risks Associated with Breast Augmentation Surgery
There are a few potential complications that are associated with breast augmentation surgery. A scar tissue capsule can occur along with effects like changes in localized sensation such as numbness in the breast or nipple. Patients may have to undergo an additional breast implant procedure to address an unwanted sense of firmness, elevation, distortion, or discomfort. About 12.5% of women who undergo breast implant surgery in the United States experience capsular contracture in one or both breasts. Choosing an experienced breast augmentation plastic surgeon will limit the likelihood of these side effects.
The Right Plastic Surgeon, the Right Breast Implants, and the Right Individualized Options
Every patient has a unique personal history and goal for a successful breast augmentation procedure. Some patients want to recover from a traumatic experience like a mastectomy or lumpectomy (in this case, considered breast reconstruction). Some patients want to improve their appearance, self-image, and confidence. The right plastic surgeon will work with you to meet your individualized needs. There is no right or wrong reason to get the breast implant surgery you deserve. Work with an experienced plastic surgeon who will get the breast implant surgery results you want.
Decisions you’ll have to make with your cosmetic surgeon before you undergo breast augmentation include:
1. The incision location: Options include the base of the breast (inframammary fold), the lower edge of the areola, the upper edge of the areola, or the armpit.
2. Length of incision: Can vary from 2 – 6.5 cms.
3. Implant insertion with regard to the chest muscles (called the ‘pectoralis major’) and breast tissue: In front or behind the chest muscle.
4. The texture of the implant: Smooth or textured. A recent 2017 study demonstrated that contoured, textured, and teardrop-shaped implants provide no benefit in terms of visible appearance over round breast implants. Further, in view of the above and recent concern over the formation of capsular lymphoma from implantation with texturing (see below), Dr. Moulton Barrett prefers to use smooth round implants.
5. The diameter of the implant: Wide and flat, narrow and round, or in between.
6. The volume of the implant.
7. The implant material: Saline, silicone gel, or gummy bear.
8. Surgical pain control using local anesthesia: Short-acting (1-2 hours), long-acting (4-5 hours ), or time-released (3-4 days ). General Anesthesia is the preferred method for surgery at a fully accredited Surgery Center: Staffed with a Board Certified Anesthesiologist and a full time / non per diem circulating and scrub nurse in the operating room suite.
9. Type of implant warranty: Duration of the warranty and financial amount of reimbursement for additional breast surgery to replace the implant and specifically A) whether or not replacement of the breast implant on the other side is covered if there is a deflation of one, and B) will a new breast implant (one or both sides) be given at no cost if there is a capsular contracture around the breast implant that will require revision surgery.
10. Breast augmentation cost: What is the out-of-pocket cost to the patient for the anesthesia fee and facility fee for implant replacement surgery.
2022 FDA Requirements for Breast Augmentation
Effective as of 11/26/2021, there are new requirements and information that must be provided by your plastic surgeon to you for all implants including saline implants:
1. You must be provided a patient information booklet specific to the implant which will be used for yourself
2. A ‘Patient Decision Checklist’ of risks, benefits, and other information should be reviewed with you by your plastic surgeon. Here are the respective forms that patients are requested to sign to confirm that informed consent has been obtained prior to breast implant surgery for the four companies that are currently licensed for breast implant sale in the United States:
-
3. You should be made aware that screening for silent rupture of a silicone implant is recommended 5-6 years after implantation and then every 2-3 years thereafter with the use of high-resolution ultrasound (HRUS) or MRI imaging.
4. You must sign that the above has taken place
5. A copy of the signed checklist should be given to you
6. Improved labelling of each implant will have been implemented by the manufacturer for later identification if needed
Breast Implant Associated Large Cell Anaplastic Lymphoma (BI-LCL)
Dr. Rex Moulton-Barrett is aware of the recent concern regarding the possible formation of a rare form of lymphoma within the capsule of breast implants. As of December 2021, there have been a total of 733 reported cases of anaplastic large cell lymphoma arising within the scar tissue capsule of breast implants in the 5-10 million women who have undergone this breast implant procedure worldwide. The incidence of this condition varies from as great as 1.79 per 1000 patients using textured Allergan implants to as little as 1 per 50,000 with the use of Mentor Siltex implants. It is for this reason textured silicone implants have been removed from clinical use in 2019 with the exception of temporary breast expansion which required contouring as part of breast reconstruction. There have been 9 reported deaths as a result of this tumor.
The median time from implant surgery to clinical diagnosis is 8 years. Of the reported cases involving implants, 60% have been following silicone gel implantation and 40% after saline, with 11% occurring with smooth-walled and 89% with textured-walled implants. Symptoms and signs most commonly would appear some years after implantation, with the finding of an asymptomatic mass within the wall or adjacent to the silicone breast implant capsule at the time of either changing the implant or removing the capsule or the formation of a new onset capsular contracture with or without the finding of a seroma (fluid collection) around the breast implant. The latter may be associated with the implant pocket enlargement and/or the sense of fullness/tension of the affected side of the chest wall containing the implant.
In 83% of the cases, the disease is confined to the capsule of the implant, with a 91% 5-year survival rate and BI-LCL accounting for just 1% of all cases of non-Hodgkins lymphoma. The current recommendation by the FDA is not to remove textured or smooth breast implants at this time. For additional information click here.
Breast Implant Warranty Information
The latest warranty information is an increasingly complicated subject. The variables include saline versus silicone gel, rupture versus capsular contracture, and year of implantation which may be quite specific to the actual implant company. To understand what warranty exists for a particular implant, please click for more detailed implant warranty information.
Frequently Asked Questions Before Getting a Breast Augmentation Surgery
Recall of Textured Allergan Biocell Breast Implants
Allergan announced a recall option on July 24, 2019 for patients to replace BIOCELL® textured silicone breast implants with smooth implants in consultation with their plastic surgeon. Allergan will provide Allergan smooth device replacements for free. The program will run for 24 months, until July 24, 2021, and will apply to revision surgeries on or after the date of the recall announcement, July 24, 2019.
The decision to get a breast implant revision or implant removal is a personal decision between patients and their plastic surgeons and must be decided based on the appropriate discussion of benefits and risks.
As part of this program, Allergan will not provide surgical fee assistance to revision or implant removal patients. This decision is in line with the FDA’s recommendation not to remove textured implants or other types of breast implants in patients who have no symptoms of Breast Implant-Associated Anaplastic Large Cell Lymphoma (“BIA-ALCL”) due to the low risk of developing BIA-ALCL.
Patients who decide to keep their BIOCELL textured devices will continue to be covered under the NATRELLE® ConfidencePlus® warranty, which includes reimbursement for up to $1,000 in diagnostic fees and up to $7,500 in surgical fees related to diagnosing and treating BIA-ALCL.
You may initiate a replacement request under the BIOCELL Replacement Warranty by talking with your Allergan Plastic Surgery Sales representative or by contacting the Allergan Product Surveillance team prior to breast implant surgery at 1-800-624-4261.
To find out more about available treatments and breast augmentation procedures, request a consultation with Dr. Moulton-Barrett at one of his Bay Area offices. Board-certified plastic surgeon Rex Moulton-Barrett, M.D., offers advanced care and plastic surgery procedures for Bay Area residents. He has offices in Alameda and Brentwood, CA.
BREAST PROCEDURES
Our Clinics
Alameda Clinic
2070 Clinton Ave
Fourth Floor
Alameda, CA 94501
Phone (510) 864-1800
Brentwood Clinic
1280 Central Blvd
Suite J-5
Brentwood, CA 94513
Phone (925) 240-8775
Our Office Location
Alameda Office
2070 Clinton Ave,
Alameda, CA 94501
510-864-1800
Alameda Location

